The appearance, taste, or odor of water from a well or other source offers some information about obvious contamination, but chemical analysis is needed to detect most contamination in water. Obvious contaminates include silt (turbidity) and hydrogen sulfide, which can be detected by smell. As a rule, the senses will not detect impurities that cause hard water, corrode pipes, and stain sinks. Two types of tests (bacteriological and chemical) are used to assess water quality.
Bacteriological Tests
Bacteriological tests are used to determine if water is bacteriologically safe for human consumption. The tests are based on detection of a group of microorganisms (coliform bacteria) that are recognized as indicators of pollution from human or animal wastes. Coliform bacteria are found in the intestinal tracts and fecal discharges of humans and all warm-blooded animals. Anyone wanting a bacteriological test performed on their drinking water should contact the Routt County Environmental Health Department at (970) 870-5588 to obtain the specially prepared bottles and instructions for taking a water sample. It’s important to note that special techniques are required to collect samples because they can be contaminated if improper procedures are used.
Chemical Tests
Chemical tests are used to identify impurities and dissolved substances that affect water used for domestic purposes. Water begins to decrease in palatability when the amount of minerals (e.g., dissolved salts) exceeds 500 to 1000 parts per million (ppm), but this depends on the nature of the minerals. Beyond these limits, the water becomes increasingly unpalatable. The table to the right lists the constituents and parameters routinely determined with a water test by the Colorado State University Soil, Water, and Plant Testing Laboratory.
The Laboratory Report—What Do the Numbers Mean?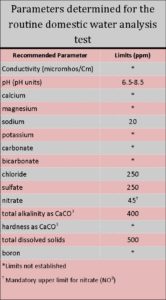
Most testing laboratories report quantities of chemical substances as weight in volumetric units such as milligrams per liter (mg/1). For all practical purposes, 1 ppm = 1 mg/l. The factors on a water analysis report are discussed below and represent the parameters considered in the evaluation of domestic water quality.
pH
pH is a measure of intensity of alkalis or acid contained in the water. Absolutely pure water has a pH value of 7.0. In Colorado the pH of well water is normally between 6.5 and 8.5. Water with pH less than 5 may cause corrosion problems because many metals become more soluble in low-pH waters.
Water with pH levels higher than 8.5 indicate that a significant amount of sodium bicarbonate may be present (see Carbonates and Bicarbonates).
Calcium (Ca) and Magnesium (Mg) cause water hardness and result from limestone-type materials in underground soil layers. Separate values are of minor concern, but they are combined for calculating hardness.
Hardness is the soap-consuming capacity of water (i.e., the more soap required to produce lather,  the harder the water). Hard water also causes greasy rings on bathtubs, greasy films on dishes or on hair after washing, and poor laundry results. Problems caused by hard water in bathing or washing may be overcome with the use of synthetic detergents or packaged “softening” compounds. The hardness of water may be removed by a water softening unit containing exchange resins, but this results in the exchange of calcium and magnesium (Ca + Mg) by sodium, so it may be a concern to people on a prescribed low-salt diet. Such water should not be used for gardens, lawns or plants. Hardness is reported as calcium carbonate (CaCO3) in mg/l.
the harder the water). Hard water also causes greasy rings on bathtubs, greasy films on dishes or on hair after washing, and poor laundry results. Problems caused by hard water in bathing or washing may be overcome with the use of synthetic detergents or packaged “softening” compounds. The hardness of water may be removed by a water softening unit containing exchange resins, but this results in the exchange of calcium and magnesium (Ca + Mg) by sodium, so it may be a concern to people on a prescribed low-salt diet. Such water should not be used for gardens, lawns or plants. Hardness is reported as calcium carbonate (CaCO3) in mg/l.
Sodium may be a health concern for people on a low-salt diet for medical reasons. Sodium can be reduced or removed by expensive treatment systems, but when calcium and magnesium are removed from water by passing through a water softener, sodium replaces it.
Potassium is an essential nutritional element, but its concentration in most drinking water is trivial and quantities seldom reach 10 mg/l.
Carbonates and bicarbonates are the major contributors to the “total alkalinity” that may be determined in a routine water test. The alkalinity of a water sample is a measure of its ability to neutralize acids. Naturally occurring levels of total alkalinity up to 400 mg/l as CaCO3 are not a health hazard. Low alkalinity can be associated with low pH values and may indicate potential for problems due to corrosion of metal in plumbing systems.
Chloride concentrations in drinking water may be important to people on low-salt diets. Most people will detect a salty taste in water containing more than 250 mg/l of chloride. Expensive treatment methods are needed to remove chloride from water.
Sulfate content in excess of 250 to 500 ppm may give water a bitter taste and have a laxative effect on people not adapted to the water. Expensive treatment methods are necessary to remove or reduce sulfate in a private water system.
Nitrate in excess of 45 mg/l (or in excess of 10 mg/l if reported as nitrate-nitrogen) is a health concern for pregnant women and infants under 6 months. High nitrate water should not be used for infant formulas or in other infant foods. Considerably higher nitrate content is apparently tolerated by most adults. Nitrates can be removed from private water supplies, but the equipment is expensive and not commonly used.
Total dissolved solids, also called “total mineral content” or “total residue,” is the total amount of material remaining after evaporation of the water. Values of less than 500 ppm are satisfactory and up to 1000 ppm can be tolerated with little effect.
Fluoride is important in the development of teeth in infants and youth. The optimum fluoride content to assist in the control of tooth decay is 0.9 to 1.5 ppm. Excessive amounts are rarely found in Colorado waters, but a concentration over 3.0 ppm may cause darkening of the tooth enamel and possibly other undesirable effects.
Iron and manganese are nuisance chemicals that cause troublesome stains and deposits on lightly colored clothes and plumbing fixtures. Iron causes yellow, red, or reddish-brown stains and deposits, while manganese stains and deposits are gray or black. Excessive amounts may also cause dark discoloration in some food and beverages and cause an unpleasant taste. Iron and manganese can be removed or reduced in a softener equipped with special resins or by small treatment systems involving aeration, filtration, and chlorination.
Taste and odor problems are difficult to solve. Some inorganic compounds may impart detectable tastes without odor. Hydrogen sulfide (rotten egg smell), when present, will impart an undesirable odor or taste. Generally, undesirable tastes may be caused by any number of organic compounds. These may be present naturally in the water, or maybe because of sewage or other surface contamination sources. These may impart disagreeable tastes and odors in minute concentrations (a few parts per billion or a few milligrams per kiloliter) and specialized chemical tests are needed to detect such small levels. Turbidity in drinking water is caused by suspended sediments from erosion and runoff discharges. The maximum contaminant level in drinking water is 1 to 5 turbidity units.
Some of the minor elements such as lead, arsenic, and molybdenum, also have recommended limits. If you have your water tested for these elements, please make sure they are below the limits and, if not, contact a professional. For example, some domestic water in Routt County has shown higher than acceptable lead contents. The Environmental Protection Agency (EPA) has determined that consumption of lead above the limits can cause mental developmental problems in adults and children.
For information regarding water that does not meet the standards contact the Routt County Extension Office or local companies that specialize in water purification.


